Environmental Bills in VA General Assembly 2017 Session Will Focus on Water Quality, Fisheries
I’ve been looking into which environmental bills and resolutions will be coming up at the 2017 Regular Session of the Virginia General Assembly. The state House and Senate will be meeting in exactly two weeks, on January 11. Several of the upcoming bills deal with water quality issues, as well as fisheries and habitat of tidal water. I’ll outline these proposed bills below, sorted via bill subject, and over the next few weeks, post updates on the states of these bills.
*Note: HB refers to House Bill followed by it’s number ID, SB refers to Senate Bill
Waters of the State, Ports, and Harbors
HB 1423/ SB 818: This bill addresses water quality in the Potomac River Watershed by designating the Virginia Department of Environmental Quality (DEQ) to 1. identify point sources when combined sewer overflow outfalls occurs, discharging untreated wastewater into the Potomac River or its tributaries and 2. gives the DEQ the responsibility of following up with owners of these discharge facilities to come up with action plans to reduce combined sewer outfalls. By doing so, the state will be acting in compliance with the Clean Water Act and the EPA’s Combined Sewer Overflow Control Policy (owners of combined sewer overflow sites must be in full compliance with this federal regulation by July 1, 2027).
A combined sewer system is when wastewater and stormwater are carried to a water treatment facility using a combined system of piping (in a separate sewer system, only wastewater is treated; stormwater flows directly into nearby streams). During periods of heavy precipitation, combined sewer systems can easily be overtaxed, leading to overflows of untreated water into streams and rivers. To my knowledge, only three major cities in Virginia still have this combined system and of these, only the city of Alexandria falls within the Potomac River Watershed. Treatment centers in this area would be subject to HB 1423/ SB 818.
Therefore, related to this last bill is SB 819, which if passed, would require the City of Alexandria to assess discharges from its Combined Sewer Outfall Number 001 (which discharges into the Potomac River) by January 2029. The City of Alexandria would have to submit this assessment to the State Water Control Board, and include an outline of actions and control technologies that must be adopted to prevent overflow discharges to the Potomac River.
HB 1454 would designate a stretch of the James River as part of the Virginia Scenic Rivers System. When the General Assembly designates a river, or sections of a river, as part of the Scenic Rivers System, it means it possesses “superior natural and scenic beauty, fish and wildlife, and historic, recreational, geologic, cultural, and other assets” (Code of Virginia, 10.1-400 Definitions). From my research, I cannot tell that this offers any kind of direct additional protection of these waters or habitats, but the designation may be important for outside conservation efforts.
Water and Sewer Systems
HB 1460 addresses the regulation of private wells, and would allow Stafford County to set standards for the construction or abandonment of private wells. Stafford County would join a growing list of state counties already able to set stronger regulations for their private wells. While this is primarily a public health issue, abandoned wells can impact groundwater, which eventually seeps into bodies of water.
Fisheries and Habitat of Tidal Waters
SB 820 would impact the Virginia Marine Resources Commission’s (VMRC) management of the menhaden industry. The bill would require the VMRC to implement the Interstate Fishery Management Plan for Atlantic menhaden and adopt regulations to manage the industry.
Atlantic menhaden can be found along the North American coast, including the Chesapeake Bay Watershed. As these fish are used for a variety of purposes, they have a long history of being overfished. Menhaden are used as bait for other fisheries (including the crabbing industry), and for fish meal and the production of fish oil. Protecting the stock of menhaden in Virginia protects a number of fishery-related industries.
Microplastics
Last month, the Washington Post published a piece on the presence of microplastics in waters all across the globe. The article featured the work of scientist Abby Barrows at Adventure Scientists, who has been collecting water samples from citizens to document the presence of microplastics.
You’ve probably heard a lot about the levels of plastic waste in our oceans. Marine birds and animals are susceptible to ingesting or strangling on large pieces of debris. What scientists are now discovering is that many of our waters have microscopic levels of plastics in them. While these microplastics pose different hazards than plastic debris, marine species could equally be as threatened. More studies need to be conducted, but there is preliminary evidence that ingesting microplastics has a negative impact on the eating habits and growth rates of marine species. Microplastics can come from a number of sources, including clothing and cosmetics. The US has already introduced a ban on microbeads in cosmetic products, with microbeads set to be phased out by 2019. However, the synthetic fibers used in the production of many of clothes (particularly fleeces), shed microplastics when washed.
Microplastics have been found throughout the world’s oceans. The Adventure Scientists are looking into how widespread pollution is. Their website features a map of all the places and watersheds where microplastics have been detected so far. I encourage you to check it out and see where there are gaps. If you plan on travelling, or if you live in an area where samples would be needed, all you have to do is sign up, read through the procedures, and take a short quiz in order to participate in sampling. They’re particularly interested in freshwater at the moment.
As G2 was already scheduled to sample in Antipoison Creek and Indian Creek earlier in November, we signed up to sample for microplastics as well. The results take about 6 to 8 weeks to come back, so we will be sure to share them as they become available!
Updates
By Kathleen
Sorry things have been silent around here on my end. I have been working “offline” on modeling and water quality analysis on several tributaries in Virginia’s Lancaster County. With input and feedback from Professor Mark Brush at VIMS, we have completed a tidal prism and two versions of a Nitrogen Loading Model (NLM) for Antipoison Creek. These NLMs show annual loads of nitrogen to the creek, but since local agricultural fields transition between corn and winter wheat at different times of the year, we have a model for each crop. I will upload a more detailed account of our findings in the next few days.
We have also begun to water sample at the neighboring Indian Creek. Dependent on time and resources, we will complete NLMs for Indian Creek, and the Corrotoman River (where water quality monitoring is conducted by an outside source and collected by the Chesapeake Bay Program).
Mid-Summer Bay Dead Zone Data Released
Earlier this summer, I reported on estimates that the Bay hypoxic zone would be smaller than average this year. The Maryland Department of Natural Resources (DNR) has just released their data for dissolved oxygen levels for July and early August in the main stem of the Bay (see this report). While the hypoxic area in August was about average, July measurements exceeded estimates.
To refresh, hypoxic zones are areas where dissolved oxygen levels fall below 2 milligrams per liter. Anoxic zones, which have not occurred this year, are areas where oxygen levels fall below 0.2 milligrams per liter. Hypoxic and anoxic zones are commonly referred to as dead zones, as low oxygen levels make it difficult for marine species to live in these areas.
Earlier estimates, made by a team of scientists from UMCES, the University of Michigan, NOAA and USGS, were calculated from spring rainfall and nutrient loading data from major Bay tributaries, such as the Susquehanna and Potomac Rivers. Spring rainfall, which carries nutrient runoff into the main stem of the Bay, is a major indicator of how big or small dead zones may be later in the year.
Measurements of dissolved oxygen are then taken throughout the summer months. Data is collected by the DNR, and compared to long-term averages (1985-2015).
July dissolved oxygen levels, and the subsequent hypoxic zone area, were higher than average. The hypoxic zone grew to 1.65 cubic miles last month, which was the seventh largest hypoxic zone on record in the Bay. (The July long-term average is 1.29 cubic miles). Early August levels were closer to estimates; the August 2016 (Aug 8-10) hypoxic zone measured 1.15 cubic miles, compared to the 1.13 cubic mile average.
Dissolved oxygen is measured with various other water quality indicators, which can give scientists a better idea of what causes average or above average dead zones. This year, scientists measured higher salinity in the Bay in June through early August. Higher salinity is indicative of less rainfall and less nutrient runoff. In wetter years, more nutrient runoff usually results in larger hypoxic zones. The results for early August confirm spring estimates. With the drier spring we had this year, we would expect to see average or lower than average dissolved oxygen levels, hypoxic zone areas, and salinity measurements.
But what about July? Why was the hypoxic zone so much larger than average? Other environmental factors, such as temperature, were likely to blame. July temperatures were especially high last month. Warmer air and water temperatures result in stagnant waters; oxygen on the Bay surface is unable to mix with bottom waters, where species such blue crabs, oysters, and finfish can be found.
Monitoring of the Bay hypoxic zone concludes in September. Check back for an update in a few weeks when the late August/ early September data is released.
More on Oyster Production: Aquaculture vs. Traditional Means of Harvest
Last week I posted a review of Kate Livie’s book, Chesapeake Oysters: The Bay’s Foundation and Future. The final portion of Livie’s book is dedicated to the rise of aquaculture in the Bay watershed over the past 15- 20 years.
Aquaculture is reliant on the production of disease-resistant, sterile triploid oysters. (To read more about the production of these triploids please see this former post on a Chesapeake Bay hatchery; and this former post on a nearby oyster farm). Raised in floating cages, or in cages suspended on the Bay floor, triploids are being raised in the millions in Virginia and Maryland, revitalizing the local oyster industry. The Chesapeake oyster can once again be found in restaurants and markets across the Eastern seaboard (and sometimes even across the country).
Aquaculture represents the future of the Chesapeake oyster in this region. However, traditional forms of harvest are still taking place in the Chesapeake and its tributaries. Despite the fact that traditional harvesting methods produce a minute fraction of historic records, harvesting of reefs, both natural and manmade, is an ongoing practice in Maryland and Virginia. Some question whether harvesting of reefs should be allowed at all, when the wild oyster population remains so low. As stated previously, the current population is estimated to be at or below 1% of historic levels.
Virginia and Maryland have both attempted to increase these population levels, with limited success. State-owned sanctuaries, where harvesting is limited or forbidden all together, are a way to protect and revive the wild oyster population. A recent report from the Maryland Department of Natural Resources unsurprisingly documents a population boom among oysters in Maryland sanctuaries over the past five years, while the oyster population outside of protected areas fell. Livie’s telling of the decline of the wild Chesapeake oyster, and this recent DNR report leave us with the questions: Should harvesting be allowed in these sanctuaries? Should harvesting continue at all while population numbers remain so low? If Maryland was more open to aquaculture, would this present a solution to displaced watermen, or those seeking alternate means of employment within the industry?
These aren’t easy questions to answer. Harvesting of oyster reefs remains an economic and cultural cornerstone of several waterfront communities in the Chesapeake region. The studies that have been undertaken do not definitively prove that harvesting of oyster reefs is harming current populations. On the other hand, with population levels as low as they are, it would seem that stricter regulations could only help.
As with many environment and water quality issues in the Bay, there are no easy solutions. But it’s certainly interesting to examine all sides to the story.
A Review of Chesapeake Oysters: The Bay’s Foundation and Future
Did you know that oyster aquaculture has been practiced and debated in the Chesapeake region since the late 1800s? William K. Brooks, a researcher with the Chesapeake Zoological Laboratory, was the first to experiment with Chesapeake oyster production in a controlled environment. Brooks, funded by the state of Maryland in the 1880s, studied and promoted ways in which oysters could be farmed to prevent against overharvest of natural oyster reefs. While hugely unpopular among the public, and especially among those in the oyster industry in his time, Brooks’ findings are used today in the booming oyster aquaculture industry.
Kate Livie, the director of education at the Chesapeake Bay Maritime Museum at St. Michael’s, Maryland, shares stories such as Brooks‘ in her 2015 book, Chesapeake Oysters: The Bay’s Foundation and Future. Live gives a fascinating account of the rise and fall and current renaissance of the Chesapeake oyster industry.
Livie’s begins her book by introducing the Chesapeake oyster and its importance in this region, by using historical accounts and anthropological evidence to describe pre- and post-colonial reliance on the oyster as major source of food. Livie argues that oysters were the foundation for colonial settlement in the Chesapeake region, and continued to be a staple in the regional diet over the next century.
Livie goes on to detail the rise of oyster harvesting in the Chesapeake Bay, which became an economic and cultural cornerstone of the region. Cities and towns, like Baltimore and Crisfield, grew in size and prominence during the oyster boom of the mid- to late-1800s. Packinghouses employed thousands of workers, charged with shucking, packaging, and shipping oysters out from the Bay region to other parts of the country.
Industrialization and advances in technology allowed watermen and packinghouses to produce, package, and ship out oysters at an unprecedented rate. They were able to do so as national demand for oysters rose. Chicago, New York, and New England were major consumers of Chesapeake oysters, as many of these places had wiped out their own native oyster beds. As a great number of packinghouses sprang up in the Chesapeake watershed, Livie paints a vivid picture of the efforts these places used to stand out. Unique and colorful cans, trade cards and advertisements for Chesapeake oysters could be found in restaurants and markets along the Eastern seaboard.
With the massive scale of oyster production going on in the nineteenth and early twentieth centuries, watermen adopted new methods, such as dredging, to harvest oyster reefs. Dredging and overharvesting led to the depletion of natural reefs and major reductions of stock. Dredging caused vertical reefs to become horizontal in structure, and made oysters more susceptible to sedimentation, and disease. This susceptibility was no more apparent than in the second half of the twentieth century, when the diseases MSX and Dermo reached the Chesapeake Bay watershed. MSX and Dermo wiped out much of -what was already- a severely depleted oyster population. Populations remain low to this day; current reports estimate that the Chesapeake oyster population is just 1% of historic levels.
Due to disease and overharvest, oyster production was a dying industry in the Chesapeake region until the early 2000s, when scientific researchers discovered a way to harvest an oyster immune to MSX and Dermo, and released this oyster to the market. The discovery of a disease-resistant triploid oyster, unable to reproduce and create spat, has resuscitated the industry. However, this revival is a complete turnaround from natural methods of harvest. Unable to form natural reefs, the triploid oyster must be farmed. Traditional oyster harvesters have been reluctant to switch to aquaculture, especially in Maryland where an aversion and distrust of oyster farming goes back to the days of William K. Brooks.
Virginia on the other hand, has been much more open to oyster aquaculture, historically and with the recent emergence of the disease-resistant triploid. While farming has had a slow start in Maryland, dozens of private oyster farming businesses popped up over the past ten years in Virginia (54 farms registered as of 2013). Outside of farming, hundreds of individuals throughout Virginia raise not-for-profit oysters in floating cages. On this explosion of aquaculture in Virginia, Livie states that from the time VIMS began tracking aquaculture, “Hatchery-produced seed plantings grew from 6.2 million in 2005 to 138 million in 2014- an increase of over 2,125 percent in less than a decade.”
If you want to read more about the emergence of aquaculture in the Chesapeake Bay watershed, or learn more about the history of the oyster in this region, pick up Livie’s book. It’s a great read.
[Livie, Kate. Chesapeake Oysters: The Bay’s Foundation and Future. Charleston: American Palate, 2015.]
Water Quality Analysis of Antipoison Creek
We have been collecting water quality data on Antipoison Creek for about 11 months now, sampling for nitrate-nitrites (NO23), phosphates (PO4), ammonium (NH4), and chlorophyll. Our data collection extends from last September to this May (with sampling ongoing). Last month, Gary, Libby and I visited with Dr. Lora Harris, an associate professor at the University of Maryland’s Center for Environmental Science (UMCES), who specializes in systems ecology and ecosystem modeling, to get input on how to analyze our water quality data.
Dr. Harris gave us several suggestions, one of which was to create a Nitrogen Loading Model (NLM) for Antipoison Creek, to determine if the creek is primarily fed by nitrogen coming in from the Chesapeake Bay, or from the watershed (land-based activity and runoff, groundwater leaching, and atmospheric deposition). Harris described the NLM as a type of box model, showing inputs and outputs in and from a watershed. (See Figure 1, from the NLM instruction manual below).

The Nitrogen Loading Model, created by Mark J. Brush (VIMS), Lora A. Harris (UMCES), Juliette C. Giordano (VIMS), and Joanna K. York (UDEL) allows a user to put in data on land use and activity in a specific watershed- for us, Antipoison Creek. The model pulls in miscellaneous information on the watershed, such as atmospheric deposition, loads from point sources, and surface area of the creek. It also pulls in data on non-agricultural and agricultural land cover, as well as the fertilization rate and agricultural yield of each crop grown in the watershed.
Using a mix of applications including a GIS operating system (QGIS) and GoogleEarth, as well as a number of sources on Antipoison Creek and Lancaster County, we came up with estimates for the needed parameters. Once all the inputs were entered, and the model generated kilograms of nitrogen for groundwater input, wastewater input, direct atmospheric deposition onto water surface, and a total annual load of nitrogen (kg) entering the watershed, we contacted Dr. Brush to discuss our procedure and results.
Now that we have an annual watershed load for nitrogen to the estuary, we must determine if Antipoison Creek has a greater flux of nitrogen coming from the watershed and emptying out into the Bay, or if the Bay is primarily feeding the creek with an influx of nitrogen.
We will be working with Dr. Brush on this next step, as well as a more in-depth paper to share our work on the NLM model.
Source: Brush, M.J., L.A. Harris, J.C.P. Giordano, and J.K. York. 2015. Delmarva Coastal Bays Nitrogen Loading Model. Virginia Institute of Marine Science, Gloucester Point, VA. Available at: http://www.vims.edu/research/departments/bio/programs/semp/models/index.php.
The Impact of Catch Share Programs on Chesapeake Fish Landings
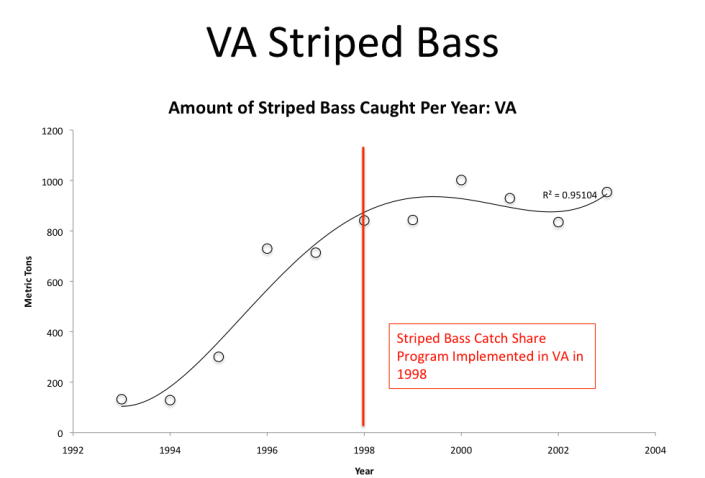
Last week, Libby introduced catch share programs, shared their pros and cons, and briefly outlined existing programs in the Chesapeake Bay region. As a continuation of her previous report, using data from NOAA and the Environmental Defense Fund, she graphed Virginia and Maryland fish landings for several species over a ten-to-twelve-year period. The data for these fish- striped bass, black sea bass, and summer flounder- shows numbers for fish landings both five-to-six years before a catch share program was implemented in each state for each species, and the five years following.
In her former piece, Libby stated that populations of striped bass in the Chesapeake had risen since catch share programs were implemented in Virginia and Maryland. Our first graph for striped bass landings in Virginia (1992 -2004) seems to correlate. Since catch share programs for striped bass were implemented in the Chesapeake region in 1998, both populations and fish landings (in metric tons) for striped bass have increased.
Catch sharing was implemented in Maryland for summer flounder in 2005. The immediate year after implementation, landings decreased, but for the next four years, landings followed an upward trend. It would seem that the catch share program has had a positive impact on harvests for the summer flounder.

Catch share programs do not seem to be having a positive effect on all Chesapeake finfish species, however – at least in terms of harvest numbers. Fish landings for black sea bass (1998-2010) decreased in both Virginia and Maryland after catch share programs were implemented in both states in 2004. It is difficult to tell if catch sharing has been a significant cause for this decline. Landings were decreasing prior to 2004, and continued to do so afterwards. It could be that a combination of factors contributed to a decline in black sea bass landings.


Catch Shares in the Chesapeake Bay and Beyond
By Libby Warner
On our visit to Antipoison Creek, we met with a fisherman who spoke somewhat highly of catch share programs. Afterwards, I investigated the purpose of catch share programs as well as the pros and cons of their implementation.
Catch share programs were originally developed in the United States to promote the economic and environmental stability of fisheries. In 1976, the federal government officially acknowledged that overfishing was a national issue and passed the Magnuson Steven’s Act. This created regional councils to manage fishing practices in different parts of the country. The act was first amended in 1996 with the Sustainable Fisheries Act. This amendment limited fisheries to only fishing during certain times of the year, a method referred to as derby fishing. Fishermen would fish as much as they could during the limited time frame, which resulted in increases in overfishing, illegal poaching, and competition and conflict between recreational and commercial fishermen. There were also several negative economic impacts associated with derby fishing. In 2006, the Magnuson Steven’s Act was amended with the Fishery Conservation and Management Reauthorization Act, which installed various catch share programs throughout the country.
In regions with catch share programs, including the Chesapeake Bay, a total maximum number of allowable catch is instated per annum for a particular species of fish. Individual fishermen and companies are then assigned a TAC, or total allowable catch, a quota based on the number of fish each company caught in the past. Many question the fairness of quotas decided in this fashion. However, the incentive of catch share programs is to limit the amount of fish caught overall rather than limiting the time of year fish can be caught.
In addition to limiting total catch, there are a number of benefits associated with catch share programs. For instance, since the implementation of catch share programs, the Chesapeake Bay, along with other bodies of water throughout the country, have seen a decrease in the number of discards and ghost fish, (fish that get caught in equipment and either die or are lost). There have also been improvements in fishermen safety. During the derby fishing days, fishing was one of the most dangerous jobs in the country. Due to time constraints, fishermen would rush out to catch fish despite poor weather conditions. With the removal of time constraints, catch share programs have provided more full-time jobs, rather than part-time jobs as fishermen now have a year-round season.
Another benefit of catch share fishing is that it increases the quality of fish caught. Since companies own portions of fish stock, they are often more invested in taking care of their stock. Being free to fish any time of year results in fishermen focusing on quality rather than quantity of fish caught. This is especially beneficial for the health of local people who rely on fish as a primary source of protein.
Whether or not catch share programs have actually made fishing more environmentally sustainable is still up for debate. Many argue that proof of success includes rebounds of fish populations across the country, including blue king crab, snow crab, Pacific coast widow rockfish, Atlantic windowpane flounder, and Gulf of Mexico red snapper. While the government has been able to increase total catch limits over time, showing proof of sustainability, many also argue that it is difficult to determine whether or not the rebounds in fish populations are a direct result of catch share programs. Many environmental activists are opposed to catch shares because it is essentially a privatization of public resources. Fishing companies are allowed to trade or sell quota, which results in armchair fishermen, (large fishing companies who sell quota to smaller companies and receive a portion of the profit). Large companies are now more capable of taking over the industry and owning most of the shares. Grassroots environmental activists use the phrase “corporate industrialization of fisheries” to describe the large fishing companies that are becoming monopolies in the fishing world. This is potentially problematic because large corporations in a capitalist society do not always rank environmental interests as a priority. Additionally, although catch share programs have resulted in a shift from part-time jobs to full-time jobs, overall, the monopoly aspect of catch shares has decreased the total number of jobs available.
Catch share programs approach conservation by focusing on individual species of fish rather than looking at aquatic ecosystems as a whole. Many environmental activists argue that the real issue is that, worldwide, there are currently approximately five times as many boats fishing as would be needed to catch a sustainable number of fish. With the implementation of catch share programs, fishermen often respond by simply moving to other parts of the world. Therefore, the total number of fish caught worldwide may not be reduced.
Catch shares are just as controversial in the Chesapeake Bay. Ten years ago, Virginia installed a catch share program in the Bay for striped bass, a population that had been overfished. After the catch share implementation, Virgina has been able to sustainably manage the striped bass population. To implement this program, NOAA provided educational sessions for striped bass fishermen to learn about catch shares and aided fishermen in the transition process from derby fishing to catch share fishing. From an original catch season of three months, the government extended the harvest period for striped bass to eleven months. The Virginia program has also allowed fisheries to be more economically successful.
Another positive result of catch share programs in Virginia is the development of “Chesapeake Catch.” Chesapeake Catch is a smartphone application developed for fishermen in the Bay, allowing them to log their catch and view data regarding other fish caught in the Bay. This has been a successful way for fishermen to come together and communicate within the industry.
Maryland took longer to implement the catch share program for striped bass; a total allowable catch was not instituted until 2014. Catch shares in both Maryland and Virginia have received criticism for putting small fishermen out of business. Throughout the Bay region, many fishermen, particularly smaller family businesses, complain that catch shares have changed their traditional fishing techniques, and allowed larger fishing corporations to control the small companies.
With striped bass being Maryland’s state fish and it’s third most valuable seafood industry, the catch share program implementation for striped bass in Maryland is very significant. Despite the complaints, since the program began, striped bass populations have improved and overfishing has declined in both Maryland and Virginia.
After exploring the different stances people take toward catch shares, and examining the pros and cons of this fishing technique, I have come to the conclusion that overfishing is an extremely complicated issue with no simple solution. It is still relatively unclear whether or not these programs are environmentally sustainable on a global scale. Catch share programs, however, seem to be effective in the Chesapeake Bay watershed. Although there are may be short-term economic downsides to catch shares, there have been ecological benefits in the Chesapeake region.
Sources:
https://www.edf.org/oceans/how-catch-shares-work-promising-solution
http://marinesciencetoday.com/2013/04/09/catch-shares-what-are-they-and-will-they-work/
https://www.youtube.com/watch?v=Sf5GlGDD40M
http://www.nmfs.noaa.gov/sfa/laws_policies/msa/
https://www.youtube.com/watch?v=oJzY5Ml1kd8https://mission-blue.org/2015/12/chesapeake-bay-suffers-from-menhaden-reduction-industry/
http://www.voanews.com/content/catch-shares-aim-sustainable-fishing/2902080.html
http://www.takepart.com/article/2013/03/18/us-fishing-rebound-it-over-small-fishermen
Osprey on Antipoison Creek

By Gary Greenwood
This past Saturday was a beautiful summer day on Antipoison Creek. As we were sitting outside enjoying the view and breeze, we would occasionally hear a loud splash just off the shore. We would look up just in time to see the osprey surface and try to gain altitude with a fresh-caught fish in his talons. We named him Sam.
There is an osprey nest on a pole not too far from our shore, close to the oyster farm. It’s on a piling, not as high above the water as it should be, but the osprey population is increasing, so they use any suitable platform they can find. Last year it was just a piece of plywood, and in a storm the nest was at risk of blowing away. So in late March my neighbor Glenn and I added some nice 6-inch walls around the edge of the plywood. We had no sooner gotten back to shore than we saw two osprey pairs checking it out. Sam and Louise won out, and proceeded to build a nice nest.
After Sam caught the fish, we could see Louise through the telescope, chattering away. Soon Sam came along with the fish, and two young chicks stuck their heads up for dinner. The feathers are all dark grey, and the necks are long. Louise would pick a piece of meat off the fish and feed it to the chicks. They aren’t ready to fly, but they do a good job eating and sleeping.
I didn’t get any good pictures on Saturday, so the next day, even though it was rainy, I went over and got a nice picture of a very wet Louise watching over her chicks. Unfortunately, the chicks weren’t sticking their heads up unless there was a fish, so you can’t see them in the picture.
- ← Previous
- 1
- 2
- 3
- …
- 18
- Next →

